
Over the past few years, the gut microbiota has been implicated in developmental disorders such as schizophrenia and autism, neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease, mood disorders such as depression, and even addiction disorders. It now seems strange that for so many decades we viewed the gut microbiota as bacteria that did us no harm but were of little benefit. This erroneous view has been radically transformed into the belief that the gut microbiota is, in effect, a virtual organ of immense importance.
What we’ve learned is that what is commonly referred to as “the brain-gut-microbiota axis” is a bidirectional system that enables gut microbes to communicate with the brain and the brain to communicate back to the gut. It may be hard to believe that the microbes in the gut collectively weigh around three pounds—the approximate weight of the adult human brain—and contain ten times the number of cells in our bodies and over 100 times as many genes as our genome.1 If the essential microbial genes were to be incorporated into our genomes, it is likely that our cells would not be large enough for the extra DNA.
Maintaining a healthy and diverse diet, exercising, and avoiding obesity as we age can have a beneficial impact on such microbes.
Many of those genes in our microbiota are important for brain development and function; they enable gut bacteria to synthesize numerous neurotransmitters and neuromodulators such as γ-aminobutyric acid (GABA), serotonin, dopamine, and short-chain fatty acids. While some of these compounds act locally in the gut, many products of the microbiota are transported widely and are necessary for the proper functioning of diverse organs. This is a two-way interaction: gut microbes are dependent on us for their nourishment. Any pathological process that reduces or increases food intake has implications for our microbes.
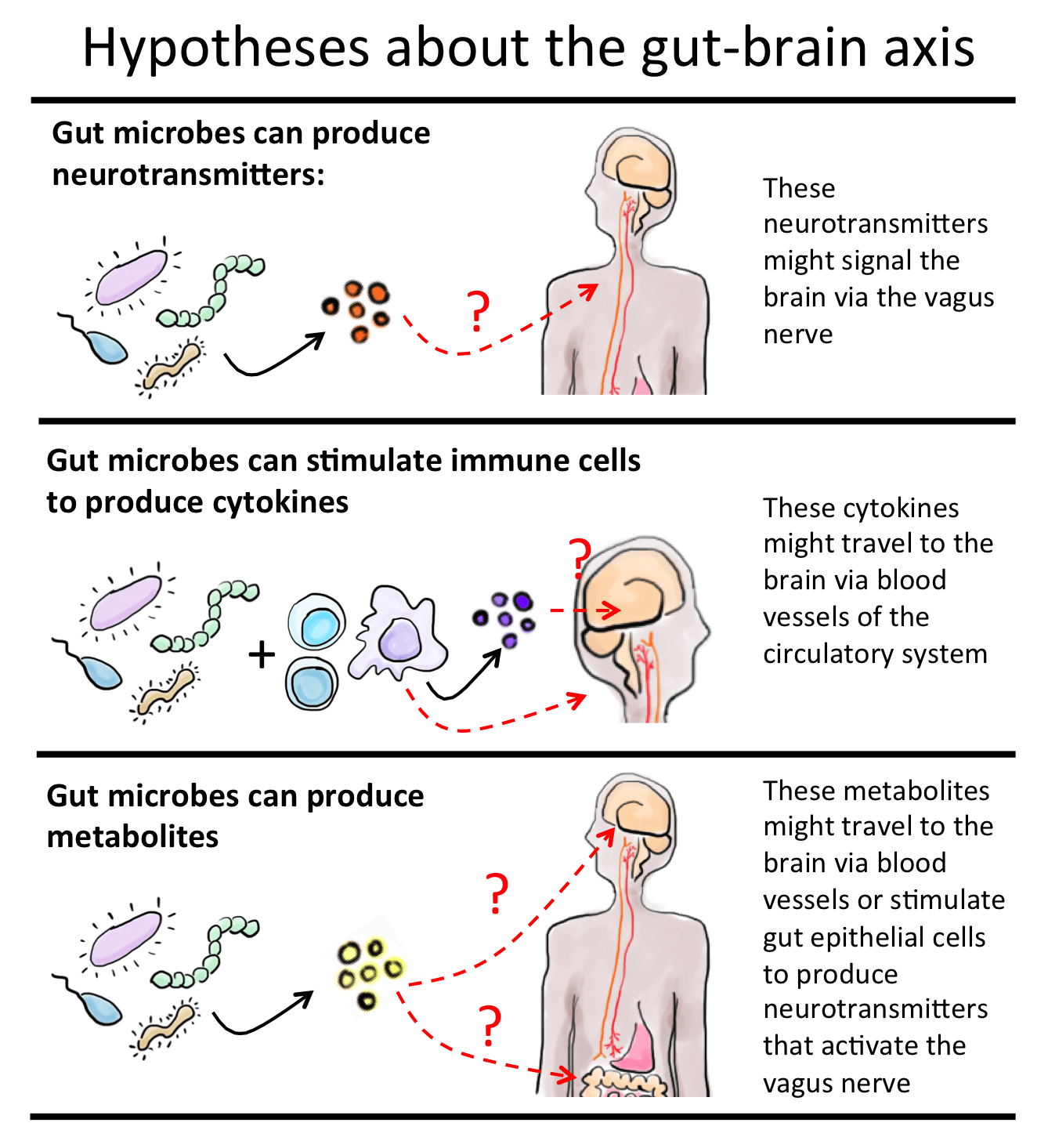
The routes of communication between gut microbes and the brain are not fully known, but they most likely include neural, endocrine, immune, and metabolic pathways.5 In particular, the long, meandering vagus nerve is thought to be a key bidirectional information pathway. Multiple chemicals are produced in the gut and travel to the brain through the blood stream. Many of these chemicals—tryptophan, leptin, and ghrelin—are produced by or have their production controlled by gut microbes and are involved in regulating mood and appetite. Cytokines, which are key immune molecules produced at the level of the gut, can travel through the blood stream and influence brain function in certain regions. One such region is the hypothalamus, where there is a deficient blood brain barrier, which in other brain regions plays a vital protective role by blocking substances in the body from entering the brain. It is certain that because of this structural difference with most other brain parts, the hypothalamus can be influenced by molecules in the blood which do not gain access to the brain elsewhere.
Influential conversations around the gut-brain axis. [o]
LINKING PARKINSON'S & MICROBIOTA
Symptoms of Parkinson’s disease (PD)—the “shaking palsy”—may have been first described by the Greek physician Galen who worked in ancient Rome. But the disease is named for James Parkinson of Shoreditch in London, who provided the first detailed description of the condition in 1817.2 This debilitating neurological disorder sometimes strikes young people but is more common in seniors. Its motor symptoms–slow movements, resting tremor, rigidity, and postural instability—predominate and can have an impact on every aspect of daily activities.
It was over a century and a half after James Parkinson that the underlying pathophysiology of the condition was understood to involve neurons in the substantia nigra, a part of the brain that plays a pivotal role in reward and movement.3 These neurons synthesise and release the neurotransmitter dopamine, which is the focus of current treatment. Dopamine enhancement does not control symptoms consistently, however. Many patients with PD need improved supplemental therapies.
PD patients frequently have non-motor symptoms that may include loss of smell (olfaction), gastrointestinal problems (especially constipation), cardiovascular dysfunction, and urogenital system difficulties. At least 50 percent develop depression during the course of their illness and approximately one in three are depressed at any point in time, sometimes prior to the onset of overt motor symptoms.
It is conceivable that PD occurs because of toxins produced by the gut microbiota or because of a failure to produce key essential neuronal dopamine specific nutrients, which are required by dopamine producing cells. The microbiota that we initially develop is determined by the mode of birth delivery of the infant (vaginally or by caesarean section), diet, and exposure to antibiotics.4 It has been shown that the core microbiota of aged individuals is characteristically distinct from that of younger adults and that age-related shifts in its composition and diversity are linked to adverse health effects in the elderly. Of interest is the fact that the number of probiotic, health-benefitting Bifidobacteria in the intestines decreases with age. In contrast, healthy young babies have large numbers of Bifidobacteria. Taking probiotic Bifidobacteria does not entirely replace these decreasing bacteria as probiotics are tourists in the intestine and do not result in permanent colonization. However, it seems likely that maintaining a healthy and diverse diet, exercising, and avoiding obesity as we age can have a beneficial impact on such microbes.

New research will hopefully affirm the validity of modulating the gut microbiota as a viable therapeutic strategy for treating neurological and psychiatric disorders. [o]
LINKING PARKINSON'S AND THE VAGUS NERVE
In the development of PD, aggregation of the protein alpha-synuclein plays such an important role that many researchers consider it central to the disease process.6 Some believe that the protein—which is manufactured by microbes—originates in the gut and spreads up the vagus nerve like a prion ( a small infectious molecule formed by abnormally folded protein). Dopamine-containing neurons within the substantia nigra—the brain region apparently responsible for the motor symptoms of PD—are especially sensitive and vulnerable to the accumulation of alpha-synuclein. The protein may also play a role in the autonomic disorders of the cardiovascular and gastrointestinal systems that are seen in PD.
The risk of developing PD is reduced in individuals who have undergone vagotomy, a surgical procedure to cut the vagus nerve, which was commonly used to treat peptic ulcer disease prior to the introduction of effective medications such as H2 antagonists and proton pump inhibitors in the 1970s and 80s. A Danish study7 investigated the risk of PD in patients who underwent vagotomy and hypothesized that truncal vagotomy (which severs the entire nerve) is associated with a protective effect, whereas super-selective vagotomy (which only partially severs the nerve) has only a minimal effect. Their findings confirmed their hypothesis: When compared to the general population, risk of PD was decreased after truncal vagotomy. These epidemiological findings, which support the view that PD initially commences in the gut and not the brain, strongly implicates the vagus nerve in the disorder’s development.
Other studies provide a mechanistic explanation. Researchers have traced the upward spread of alpha-synuclein from the gut along vagal nerve fibres.6 This suggests a neuronal route for the transport of probable PD pathogens or toxins from the enteric nervous system in the gut to the brain, rather than via the bloodstream.
THE ROLE OF GUT DYSBIOSIS
The gut microbiome in PD has only recently been investigated, and researchers speculate whether microbes are altered in PD. Sheperjans and colleagues8 compared the fecal microbiomes of 72 PD patients and 72 control subjects by sequencing the bacterial 16S ribosomal RNA gene, which is used to identify specific bacteria. Associations between clinical parameters and microbiota were analysed, considering potential confounding factors. On average, the abundance of Prevotellaceae microbes (normally found in very high concentrations in the gut of vegetarians)in faeces of PD patients was reduced by 77.6 percent, compared with controls. Relative abundance of Prevotellaceae of 6.5 percent or less had 86.1 percent sensitivity and 38.9 percent specificity for PD. An analysis based on the abundance of four bacterial families and the severity of constipation identified PD patients with 66.7 percent sensitivity and 90.3 percent specificity.
Interestingly, the relative abundance of a different type of microbe, Enterobacteriaceae, was positively associated with the severity of postural instability and gait difficulty. Enterobacteriaceae can be harmless or pathogenic as in the case of salmonella. These findings suggest that the intestinal microbiome is altered in PD and is related to the motor phenotype, though one cannot conclusively dismiss the possibility that these changes are epiphenomenal. Such findings raise the exciting possibility that changes in the gut microbiota may be used as a diagnostic marker, though further large-scale studies are required.
Mazmanian’s group9 have investigated the genesis of PD features in mice genetically engineered to overexpress alpha-synuclein, which go on to develop features of PD. These mice are now one of the most widely used animal models in studying the disorder. Intriguingly, when they are raised germ-free (i.e., without gut microbes), their tendency to develop motor abnormalities is significantly reduced. When they are given a combination of short chain fatty acids, on the other hand, they show microglial activation in the brain and aggregation of alpha-synuclein with onset of motor features. These changes are inhibited by treatment with the antibiotic minocycline, which acts on a broad range of bacteria. If germ free alpha-synuclein overexpressing animals are given a humanized microbiota from a patient with PD, the emergency of pathology is far greater than those transplanting with the microbiota from a healthy subject. These findings pinpoint several potential lines of treatment, including the use of short-chain fatty acid antagonists, antibiotics, and microbiota transplantation. Time will tell if any of these potential therapeutic options prove fruitful.
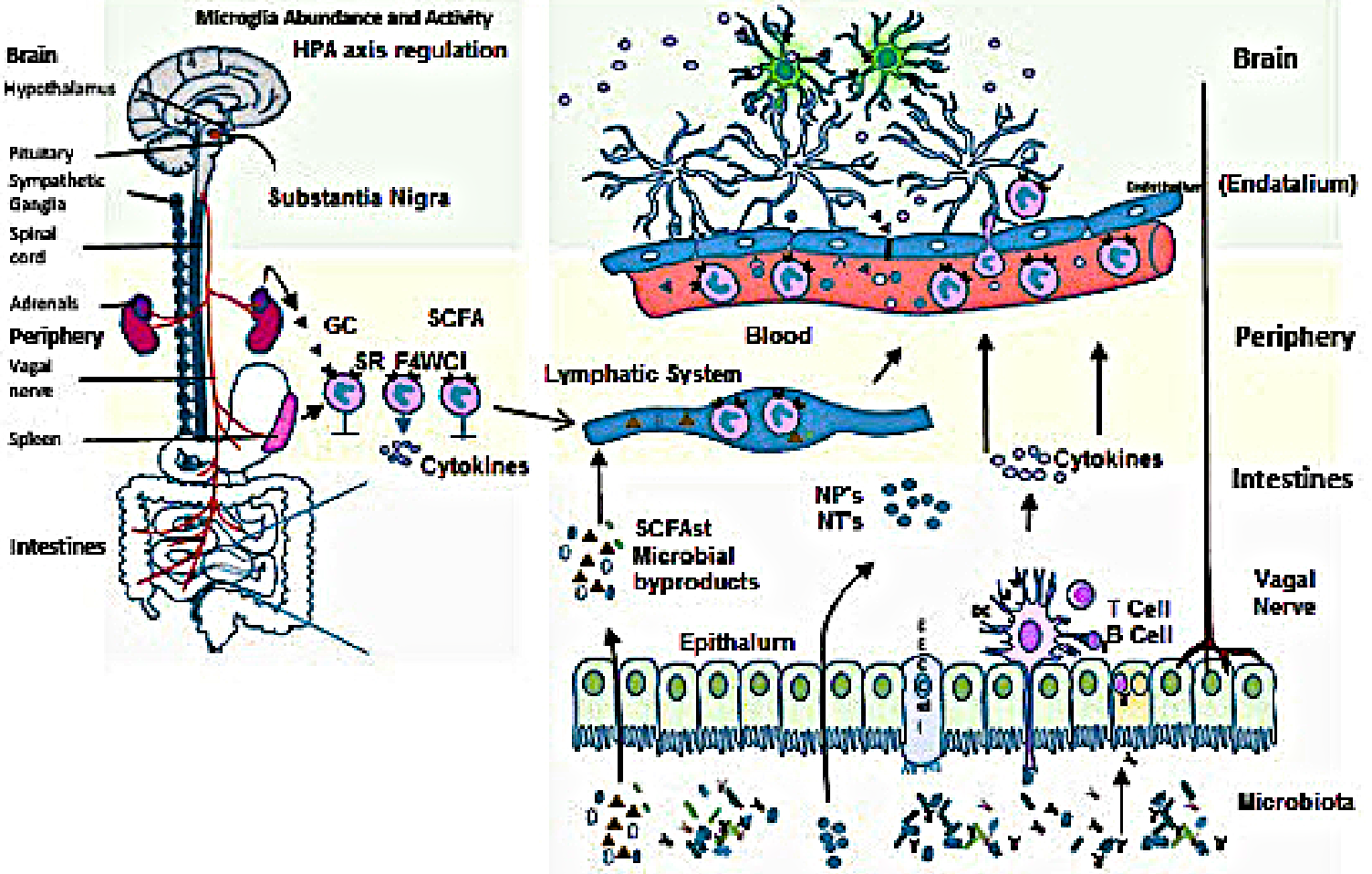
[FIGURE 1. Communication within the microbiota-gut-brain axis involves the co-ordination of multiple factors, including vagal nerve activation, cytokine production, neuropeptide/neurotransmitter release and short chain fatty acid release. When these signals penetrate the blood brain barrier, they influence the maturation and activation state of the microglia, key immune cells in the brain. Once activated, microglia play a fundamental part in immune surveillance, synaptic pruning, and clearance of debris. The HPA axis by cortisol production can in turn suppress the activation state of brain microglia, as well as influence cytokine release and trafficking of monocytes from the periphery to the brain. NB: To make this graphic viewable we have had to sacrifice image definition.]
LINKING PARKINSON'S & DEPRESSION
PD and depression frequently go hand in hand. Given the impact of PD symptoms on quality of life, it is not surprising that many patients become depressed. However, other pathophysiological aspects of PD may also render sufferers vulnerable. In a recent study, the status of non-dopamine neurons, including those that primarily express the neurotransmitters acetylcholine, norepinephrine, and serotonin, was examined in post-mortem brains of PD patients.10 In comparison with controls, PD brains showed widespread deficits of both serotonin and serotonin transporter, especially in brain areas called the caudate nucleus and the middle frontal gyrus. Significantly reduced norepinephrine levels were found in all brain regions tested, while acetylcholine transporter levels were not altered. The non-dopaminergic changes in transmitters such as serotonin and noradrenaline might well account for the high prevalence of depression in PD. While the core movement problems seen in PD are induced by a lack of dopamine, the co-morbid mood related features are likely due to changes in other neurotransmitters.
MICROBIOTA IN DEPRESSION
Recently, depression per se has also been associated with significant microbiota changes. Jang et al11 analysed faecal samples from 46 patients with major depression and 30 healthy controls. Those who were acutely depressed had higher levels of Bacteroidetes, Proteobacteria, and Actinobacteria, whereas level of Firmicutes was significantly reduced. The exact physiological implications of this are not fully understood. However, a negative correlation was observed between Faecalibacterium and the severity of depressive symptoms.
Another study, conducted at the APC Microbiome Institute,12 found an association between depression and decreased gut microbiota richness and diversity. When faecal microbiota was transplanted from depressed patients into microbiota-depleted rats, it induced behavioral features characteristic of depression, including anhedonia and anxiety-like behaviors, as well as alterations in tryptophan metabolism. This suggests that the gut microbiota may play a causal role in the development of mood disorder.
FAECAL MICROBIOTA TRANSPLANTATION (FMT) FOR PARKINSON'S?
The concept of FMT for treatment of human gastrointestinal disease was described almost 2,000 years ago by a Chinese physician, Ge Hong, who orally administered human faecal suspension to treat patients with food poisoning or severe diarrhoea. Borody et al13 noted that FMT may have been first used in veterinary medicine by the Italian anatomist Fabricius Aquapendente in the 17th century and that the first report of FMT in modern times was in the late 1950s for the treatment of pseudomembranous colitis.
Over the past few years, FMT has been increasingly used to treat resistant Clostridium difficile infection. It is remarkably effective in this application. There are obviously challenges in determining appropriate donors and which route of administration works best. Some gastroenterologists use a nasogastric route (the insertion of a plastic tube through the nose, past the throat, and down into the stomach), while other use enemas. Capsules filled with faecal material and taken orally are another option.
While Clostridium difficile is the only condition for which FMT is currently used, others are being considered. There have been case reports suggesting efficacy in PD, but it is yet to be determined whether it is a realistic therapeutic option. If gut microbes are therapeutically beneficial, then probiotics or cocktails of probiotics (polybiotics) may also prove beneficial. We await studies of these approaches.
PSYCHOBIOTICS & THE WAY FORWARD
Probiotic bacteria with a mental health benefit more generally have been termed psychobiotics. Several bacteria have been profiled pre-clinically and show promise, but large scale translational studies are required. A recent study found a Bifidobacterium longum strain to reduce anxiety levels in healthy subjects and to reduce levels of the stress hormone cortisol.14 It is tempting to speculate that psychobiotics will someday be used to treat mild forms of both depression and anxiety.
In the next few years, new research will hopefully affirm the validity of modulating the gut microbiota as a viable therapeutic strategy for treating neurological and psychiatric disorders. For example, emerging evidence supports the view that dysbiosis within the gut is the trigger for degeneration of central dopamine neurons in PD. If this proves to be the case, therapies that actually target the disease process may quickly become available, providing radical, more effective alternatives for patients. Such a breakthrough is badly needed. ũ
NOTES
1 Stilling RM, Dinan TG, Cryan JF. "Microbial genes, brain & behaviour — epigenetic regulation of the gut-brain axis." Genes Brain Behav 2014, 13(1): 69-86.
2 Donaldson IM. James Parkinson's essay on the shaking palsy. J R Coll Physicians Edinb 2015, 45(1): 84-86.
3 Jagmag SA, Tripathi N, Shukla SD, Maiti S, Khurana S. "Evaluation of Models of Parkinson's Disease." Front Neurosci 2015, 9: 503.
4 Moloney RD, Desbonnet L, Clarke G, Dinan TG, Cryan JF. "The microbiome: stress, health and disease." Mamm Genome 2014, 25(1-2): 49-74.
5 Grenham S, Clarke G, Cryan JF, Dinan TG. "Brain-gut-microbe communication in health and disease." Front Physiol 2011, 2: 94.
6 Phillips RJ, Walter GC, Wilder SL, Baronowsky EA, Powley TL. "Alpha-synuclein-immunopositive myenteric neurons and vagal preganglionic terminals: autonomic pathway implicated in Parkinson's disease?" Neuroscience 2008, 153(3): 733-750.
7 Svensson E, Horvath-Puho E, Thomsen RW, Djurhuus JC, Pedersen L, Borghammer P, et al., Does vagotomy reduce the risk of Parkinson's disease: The authors reply. Ann Neurol 2015, 78(6): 1012-1013.
8 Scheperjans F, Aho V, Pereira PA, Koskinen K, Paulin L, Pekkonen E, et al. "Gut microbiota are related to Parkinson's disease and clinical phenotype." Mov Disord 2015, 30(3): 350-358.
9 Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, et al. "Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson's Disease." Cell 2016, 167(6): 1469-1480 e1412.
10 Buddhala C, Loftin SK, Kuley BM, Cairns NJ, Campbell MC, Perlmutter JS, et al. Dopaminergic, serotonergic, and noradrenergic deficits in Parkinson disease. Ann Clin Transl Neurol 2015, 2(10): 949-959.
11 Jiang H, Ling Z, Zhang Y, Mao H, Ma Z, Yin Y, et al. "Altered fecal microbiota composition in patients with major depressive disorder." Brain Behav Immun 2015, 48: 186-194.
12 Kelly JR, Borre Y, C OB, Patterson E, El Aidy S, Deane J, et al. "Transferring the blues: Depression-associated gut microbiota induces neurobehavioural changes in the rat." J Psychiatr Res 2016, 82: 109-118.
13 Borody TJ, Brandt LJ, Paramsothy S, Agrawal G. "Fecal microbiota transplantation: a new standard treatment option for Clostridium difficile infection." Expert Rev Anti Infect Ther 2013, 11(5): 447-449.
14 Allen AP, Hutch W, Borre YE, Kennedy PJ, Temko A, Boylan G, et al. "Bifidobacterium longum 1714 as a translational psychobiotic: modulation of stress, electrophysiology and neurocognition in healthy volunteers." Transl Psychiatry 2016, 6(11): e939.
This article, reprinted here with permission of the Dana Foundation, appeared on April 30, 2017 in its online publication, Cerebrum.
PODCAST. Included with this article is a supplementary interview by Cerebrum editor Bill Glovin with Ted Dinan, which further explains the article above, as well as how diversity of diet, probiotics and other factors influence the production of 'good bacteria' in the digestive tract and consequent overall health.

TED DINAN, M.D., Ph.D., is professor of psychiatry at University College Cork in Ireland, and a principal investigator in the APC Microbiome Institute. He has published over 400 papers and numerous books on pharmacology and neurobiology and is on the editorial boards of several journals. Dinan has worked in research laboratories on both sides of the Atlantic, obtained a Ph.D. in pharmacology from the University of London, and was awarded the Melvin Ramsey Prize for research into the biology of stress.
JOHN F. CRYAN, Ph.D., is professor and chair in the Department of Anatomy and Neuroscience, University College Cork in Ireland, and a principal investigator in the APC Microbiome Institute. Cryan has published over 330 papers and has edited three books, including Microbial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease (2014). His awards include UCC Researcher of the Year in 2012 and the University of Utrecht Award for Excellence in Pharmaceutical Research in 2013.

Comments
My husband was diagnosed of…
My husband was diagnosed of Parkinsons disease two years ago, when he was 59. He had a stooped posture, tremors, right arm does not move and also a pulsating feeling in his body. He was placed on Senemet for 8 months and then Siferol was introduced and replaced the Senemet, during this time span he was also diagnosed with dementia. He started having hallucinations, lost touch with. Suspecting it was the medication I took him off Siferol (with the doctor’s knowledge) and put him on Parkinson’s Disease natural herbal formula, his symptoms totally declined with three weeks use — Tree of Health Parkinson's disease natural herbal formula. He is now almost 61 and doing very well. The disease is totally reversed.
Add new comment